1. Introduction: The Billion-Dollar Countdown
In the upper echelons of pharmaceutical leadership, the „Patent Cliff“ is frequently discussed as a looming catastrophe. However, for the veteran strategist, Loss of Exclusivity (LOE) is not a crisis—it is a predictable milestone. Treating LOE as an unforeseen disaster is a failure of foresight; instead, it must be viewed as a strategic window that requires a fundamental shift in brand narrative years before the first generic enters the fray. This transition from protected monopoly to a competitive landscape necessitates a pivot from reactive defense to „Drug Life Optimization.“
The financial gravity of this milestone is immense. Between 2022 and 2030, over 190 products will face LOE, placing approximately $300 billion in sales at risk by 2028. The erosion is not just deep; it is instantaneous, particularly for small-molecule assets.
| Metric | Typical Impact/Range |
|---|---|
| Revenue Decline (Branded Drug) | Up to 80–90% within the first year |
| Market Share Loss (Branded Drug) | Up to 73% within just two weeks of LOE |
| Price Reduction (Generic Entry) | 20–85% lower than branded counterpart |
| Industry-Wide Sales at Risk | ~$300 billion (2023–2028) |
Critically, the „cliff“ looks different depending on the molecule’s complexity. While small molecules face sharp, immediate erosion, biologics—exemplified by AbbVie’s Humira—experience a more prolonged erosion due to manufacturing complexities and regulatory hurdles for biosimilars. Regardless of the molecule type, these staggering figures demand that we move beyond „managing decline“ and instead integrate lifecycle optimization into the very core of our commercial strategy.
2. The Three Acts of Drug Life Optimization
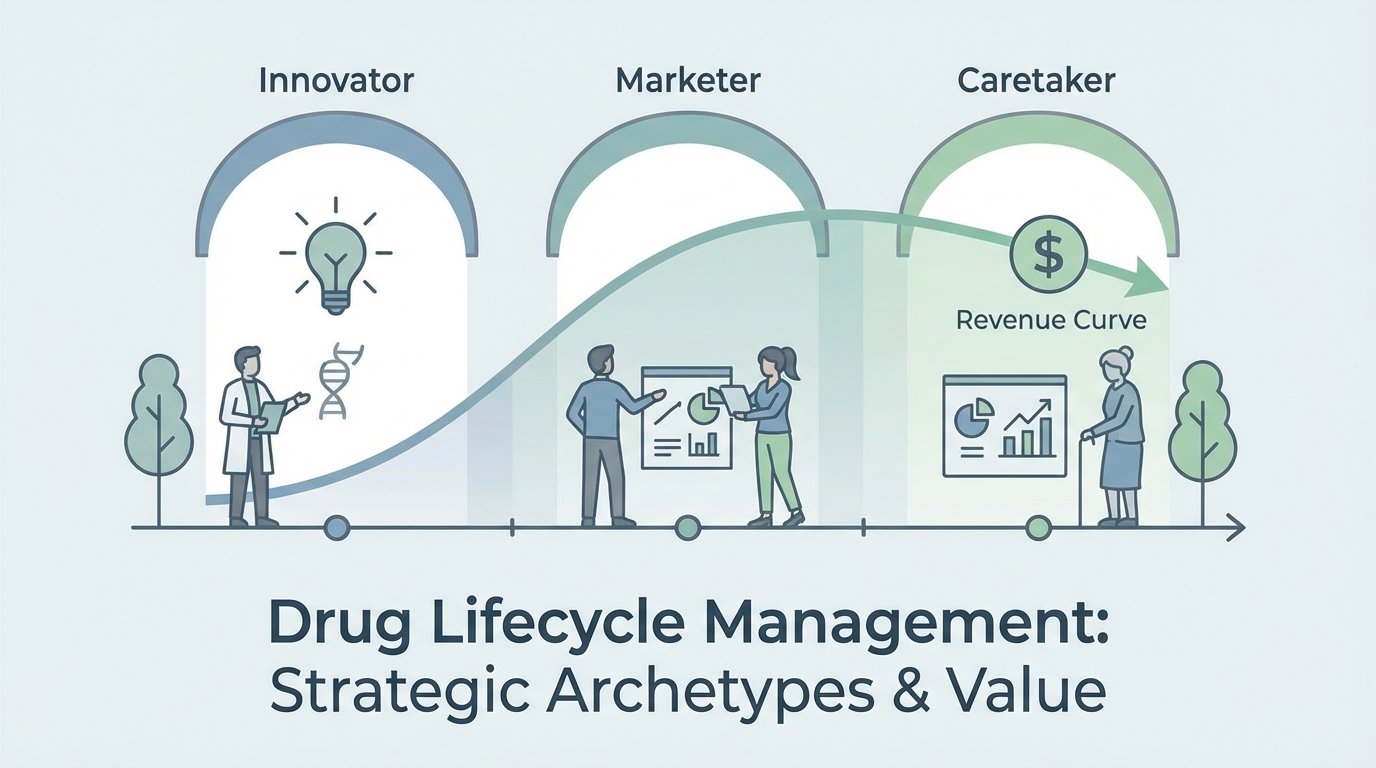
A drug does not have a single commercial life; it evolves through three distinct strategic acts. Organizations that fail to recognize these shifts often find themselves applying „Innovator“ logic to „Caretaker“ problems, leading to value destruction and internal friction.
- Early Development (The Discovery Phase): This phase often lasts a decade or longer. While this „bench to bedside“ period is the foundation, it drastically reduces the „effective market life“ to a mere 10–15 years. Strategic decisions here are influenced by a diverse web of stakeholders—regulators and payers—who dictate the future parameters of access.
- Mid-Life (The High-Stakes Growth Phase): Following launch, the objective is rapid market share acquisition and peak sales volume while under the umbrella of patent protection.
- Late Post-Patent (The Maturity and Decline Phase): As patent expiry emerges, the brand enters a defensive posture. The goal is no longer just expansion, but the retention of a „long-tail“ revenue stream against aggressive generic competitors.
As an asset moves through these stages, a company’s identity must shift across three archetypes: The Innovator, focused on technology and lead-time reduction; The Marketer, emphasizing efficiency to maintain share; and finally, The Caretaker, managing the asset’s sunset for maximum remaining value. This transition often causes significant cultural friction; an organization built on the high-risk, high-reward „Innovator“ mindset must learn the disciplined, cost-optimized „Caretaker“ model to preserve profitability. Mastering this evolution is the precursor to building a defensible pre-market foundation.
3. Foundation and Launch: Building the „Web of Protection“
Integrated lifecycle management begins 2–3 years before a drug ever hits the market. This pre-launch window is where regulatory affairs and marketing must align to gather the safety and efficacy data necessary to support future „long-tail“ revenue.
The goal is to construct a „web of protection“—a multi-layered competitive moat that goes beyond a single patent. This involves a sophisticated interplay between the Primary Patent (Composition of Matter) and Secondary Patents (formulations, dosages, or methods of use). Strategists use these „incremental hurdles“ to create „patent thickets,“ often navigating the Hatch-Waxman Paragraph IV certification process as a deliberate defensive barrier.
- NCE Exclusivity: 5 years for New Chemical Entities.
- Orphan/Pediatric Exclusivity: 7 years for Orphan drugs, with an additional 6 months for pediatric studies.
- Evergreening: Securing additional protections through modified delivery or combinations.
Case Study: The AstraZeneca Nexium Strategy & Dual Branding AstraZeneca’s transition from Prilosec to Nexium is the industry paradigm for the „chiral switch.“ By isolating the S-enantiomer and securing 40+ patents, they successfully transferred 40% of their patient base to the new protected asset before the Prilosec cliff. However, the most sophisticated players also utilize Dual Branding—marketing the same molecule for different indications under different names. Merck’s finasteride is a masterclass in this: marketed as Proscar for BPH and Propecia for hair loss, allowing for distinct pricing and payer navigation. Similarly, Pfizer utilized Sildenafil as both Viagra (ED) and Revatio (PAH) to maximize value across vastly different clinical and payer landscapes.
4. The Strategic Pillars of Value Maximization
During the maturity stage, defensive strategies must be multi-layered to insulate the brand from the impending generic „Gx first“ payer attitude.
- Pillar 1: Line Extensions. Shifting volume to newly protected assets like extended-release (ER) versions (e.g., Namenda XR) or combination products (e.g., Janumet) is critical. By migrating the patient base to a line extension before LOE, you insulate that volume from generic substitution.
- Pillar 2: Rx-to-OTC Switches. Converting to over-the-counter status can increase utilization by 30%. Success depends on consumer self-diagnosis and a robust safety profile.
| Success Criteria | Strategic Challenges |
|---|---|
| Ability to self-diagnose | Low margins vs. Rx products |
| Robust safety profile | Stringent FDA data requirements |
| Strategic Timing: Launch at LOE | Vastly different economics (High Volume/Low Price) |
- Pillar 3: Pricing and the Inflation Reduction Act (IRA). Many firms implement „Surge Pricing“ 12–18 months before LOE. However, data-driven strategists know that price increases greater than 9% demonstrably lead to negative revenue impacts. Furthermore, the IRA introduces new complexity: Medicare now has the authority to negotiate prices on high-spending, single-source drugs, with specific distinctions between Medicare Part B and Part D negotiations. Strategic rebate management is now a prerequisite for navigating these managed care hurdles.
5. Competitive Intelligence: Transforming Data into Market Domination
Competitive Intelligence (CI) is no longer a support function; it is a foundational capability for portfolio management. Advanced CI allows firms to predict freedom-to-operate issues, potentially saving hundreds of millions in development.
Drawing on the Birkinshaw framework, effective management requires:
- Sensing: Real-time tracking of R&D, patent filings, and clinical registrations to identify emerging threats.
- Seizing: Mobilizing resources for Paragraph IV litigation or bolt-on acquisitions.
- Reconfiguring: Utilizing AI and Machine Learning to accelerate R&D and refine clinical trials.
By adopting a „fail fast, fail cheap“ philosophy enabled by AI, companies can replenish their pipelines more efficiently. This ensures that as one flagship product approaches its cliff, the next generation of innovation is already entering its growth phase.
6. The „Long Tail“: Medical Affairs and Social Responsibility
In the final phase of a drug’s life, Medical Affairs emerges as a strategic leader. The „MAPS 2030 Vision“ positions this function at the center of the „long tail,“ where the brand’s value resides in the Real-World Evidence (RWE) and data legacy accumulated over decades.
This shift toward Patient-Centricity means gathering data for expanded populations—such as pediatric use—long after the initial launch. Post-LOE, the value of a drug is often found in this data, not just the molecule itself. While these strategies drive sustained revenue, they also foster social change by improving patient outcomes, increasing access to safe medicines via OTC switches, and eventually paving the way for high-quality, affordable generics. A drug’s branded „end-of-life“ is merely its evolution into a foundational element of the broader healthcare ecosystem.
7. Conclusion: The Lifecycle as a Competitive Advantage
The Patent Cliff is not a disaster; it is a predictable milestone that separates reactive organizations from strategic leaders. By integrating Intellectual Property, Marketing, and Medical Affairs into a cohesive lifecycle strategy, firms can transform LOE into a springboard for the next generation of innovation.
Strategic Checklist for Marketing Managers:
- [ ] Fortify IP Thickets: Construct multi-layered patents protecting incremental innovations and secondary attributes.
- [ ] Proactive Line Extensions: Prioritize line extensions with demonstrable clinical benefits to facilitate early „volume shifting.“
- [ ] Dual Branding Assessment: Evaluate opportunities for same-molecule, different-indication branding to navigate payer landscapes.
- [ ] AI-Driven CI: Implement real-time monitoring of competitor R&D and patent filings for early „freedom-to-operate“ awareness.
- [ ] Strategic Rebate Management: Align contracts with PBM motivations to maintain access as LOE nears.
- [ ] Managed Care Navigation: Account for IRA impacts on Medicare Part B and D for high-spending assets.
- [ ] Pricing Discipline: Ensure surge pricing remains below the 9% threshold to avoid revenue erosion.
- [ ] Evaluate Rx-to-OTC: Determine if high-safety, high-utilization products are candidates for volume-driven OTC models.
Managing the drug lifecycle is the ultimate business challenge. It requires an integrated, strategic mindset that views the patent cliff as just one act in a much longer game. Those who master this game do more than just survive; they define the future of medicine.





